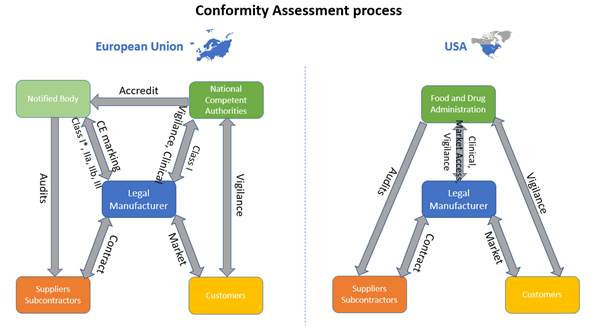
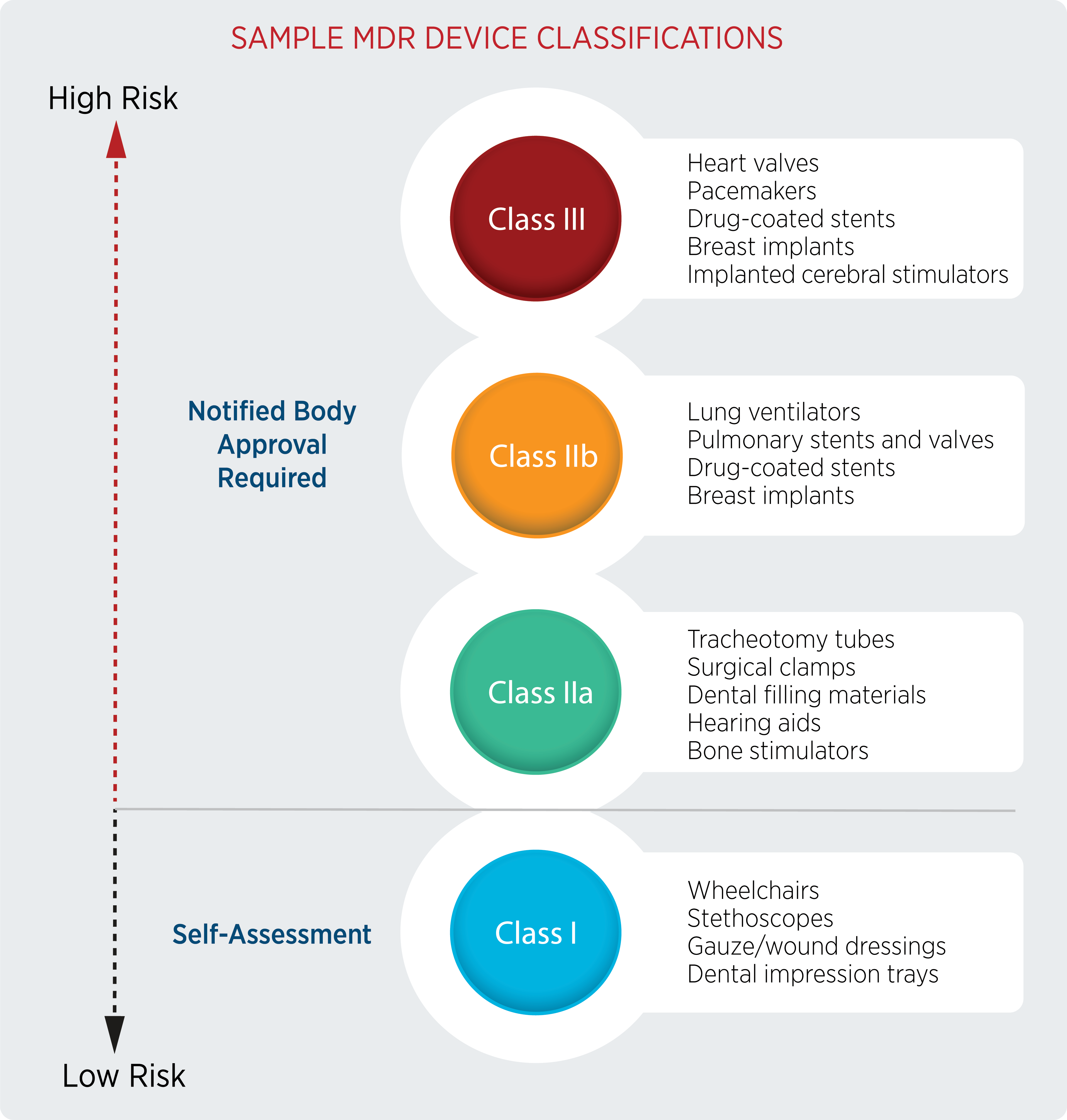
What Notified Bodies Look for When Reviewing Your Medical Device Technical Documentation Under the EU MDR – Oriel STAT A MATRIX – ELIQUENT Life Sciences Blog
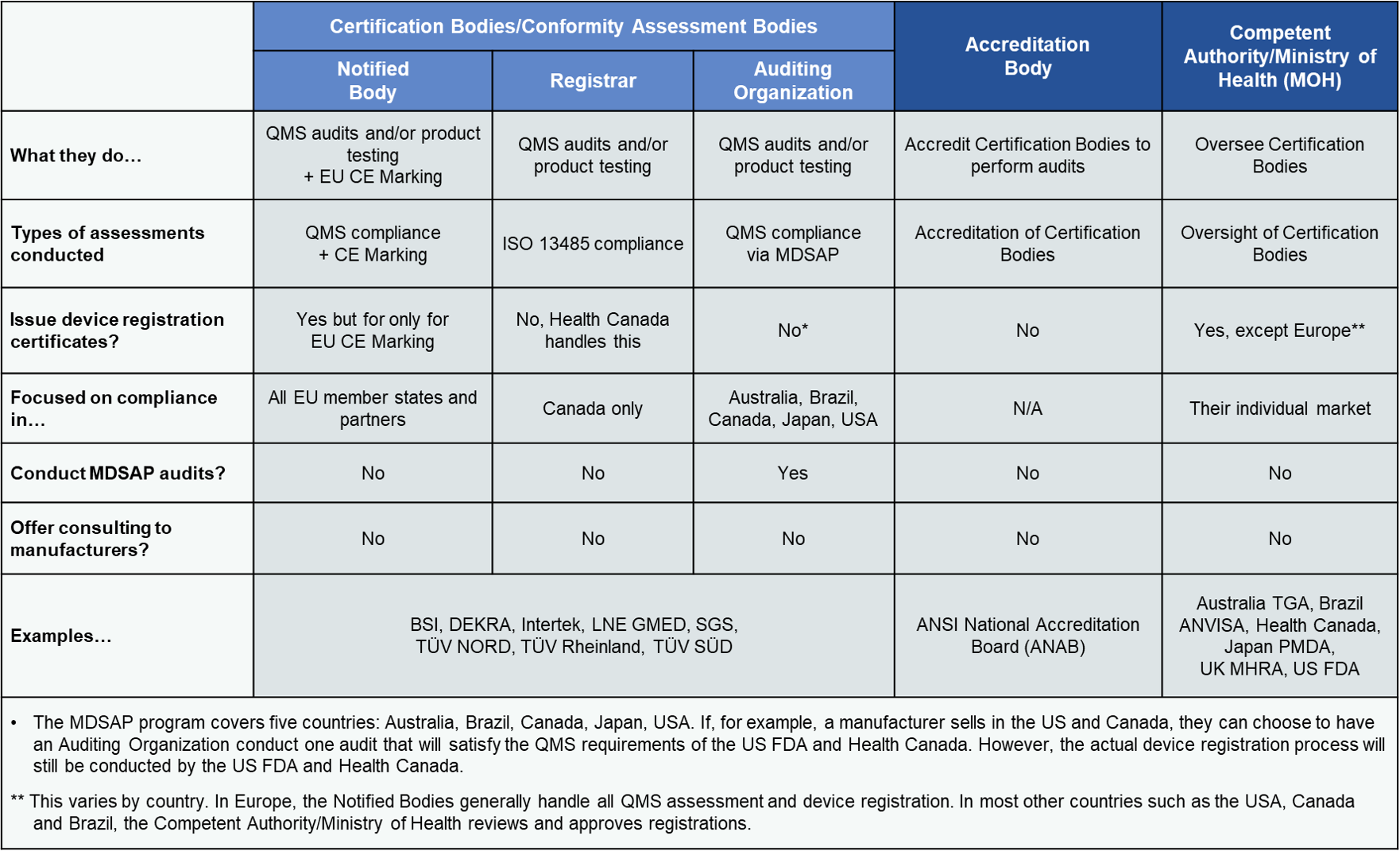
Auditing Organization (AO) versus Notified Body (NB) versus Registrar. What's the difference? – Oriel STAT A MATRIX – ELIQUENT Life Sciences Blog