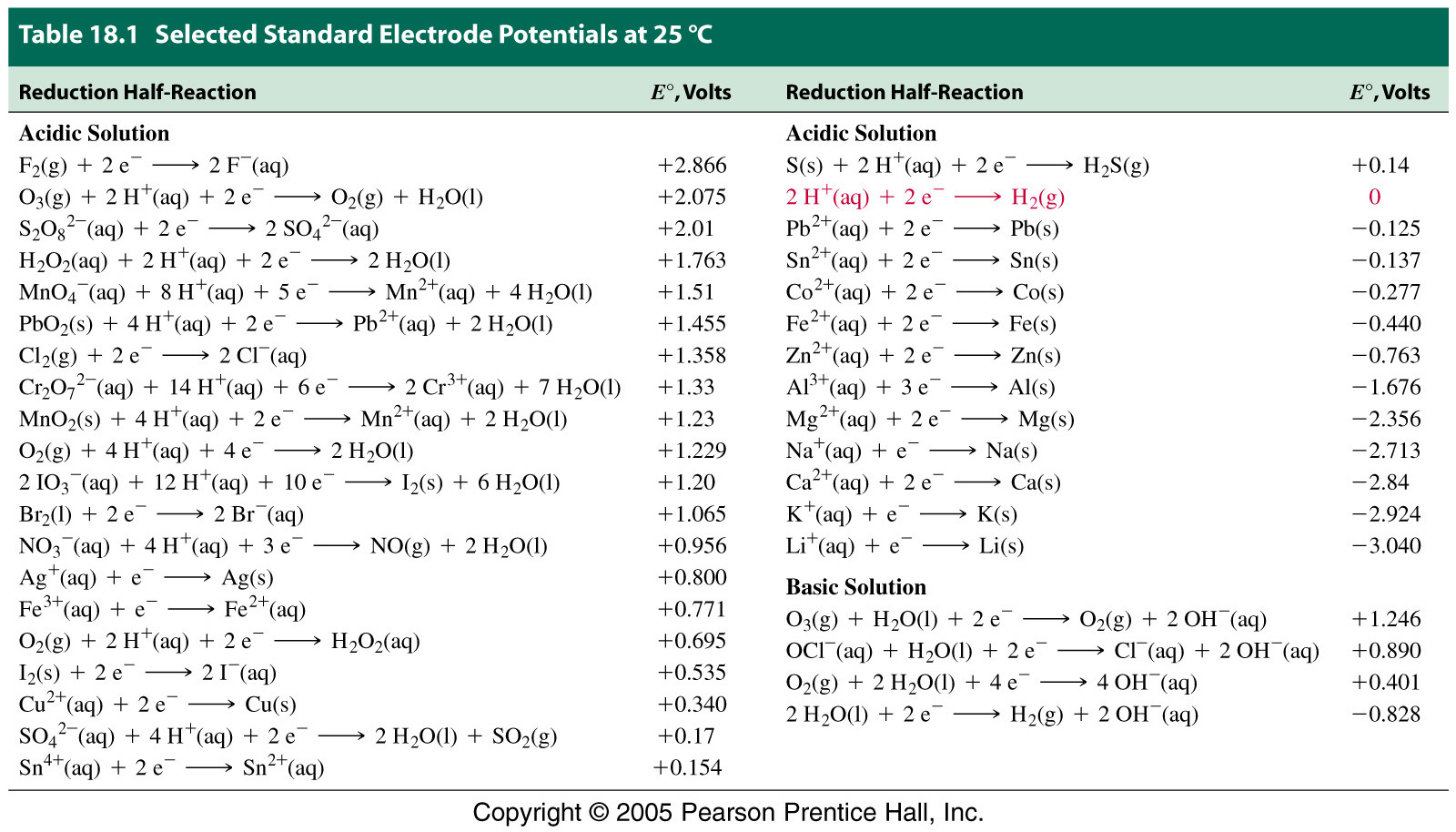
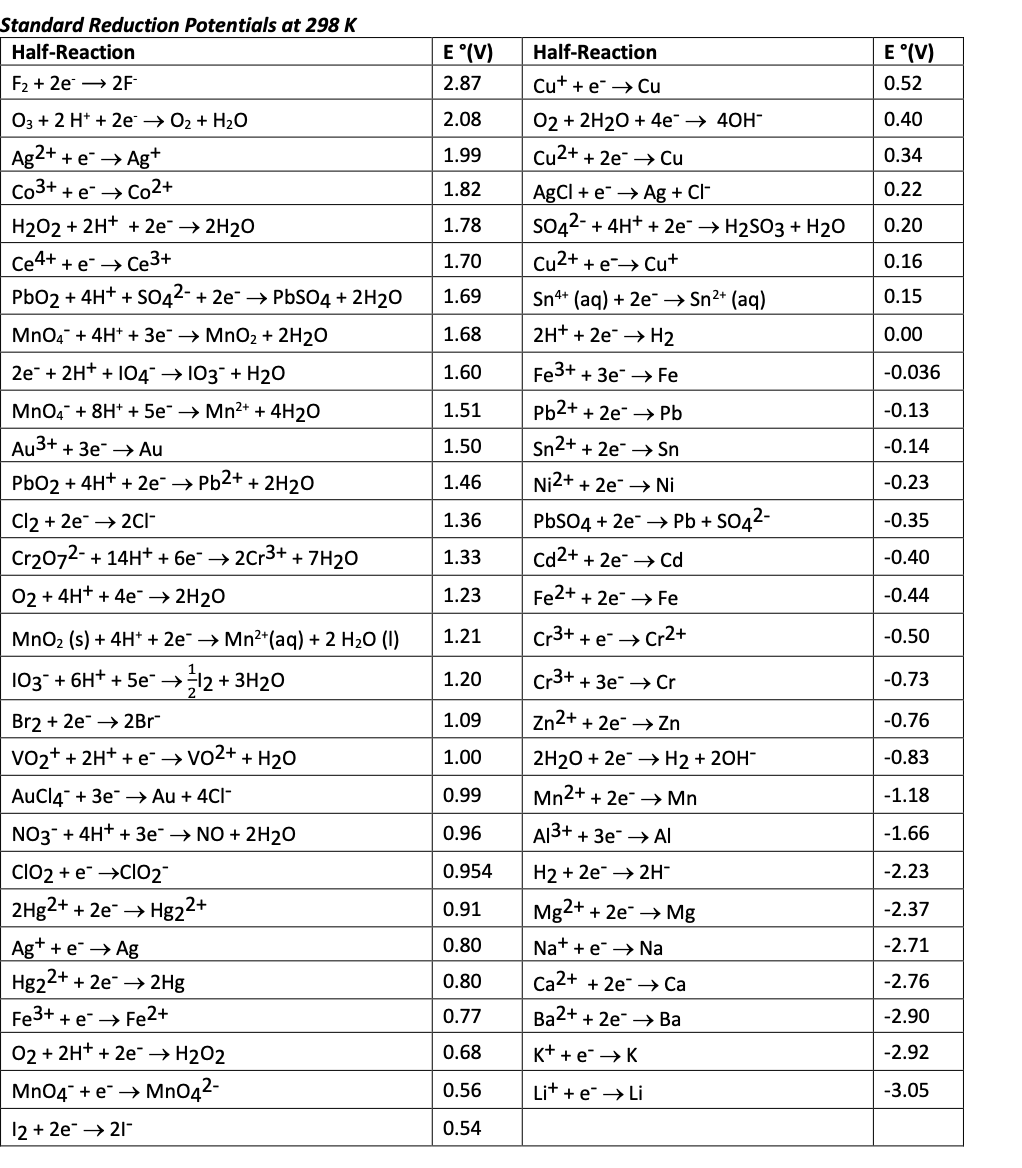
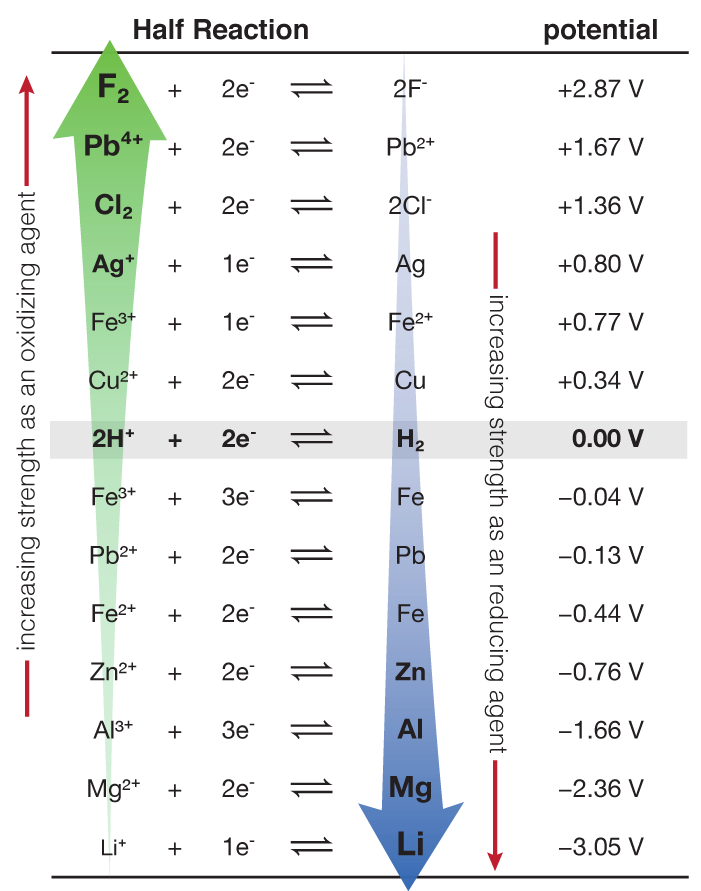
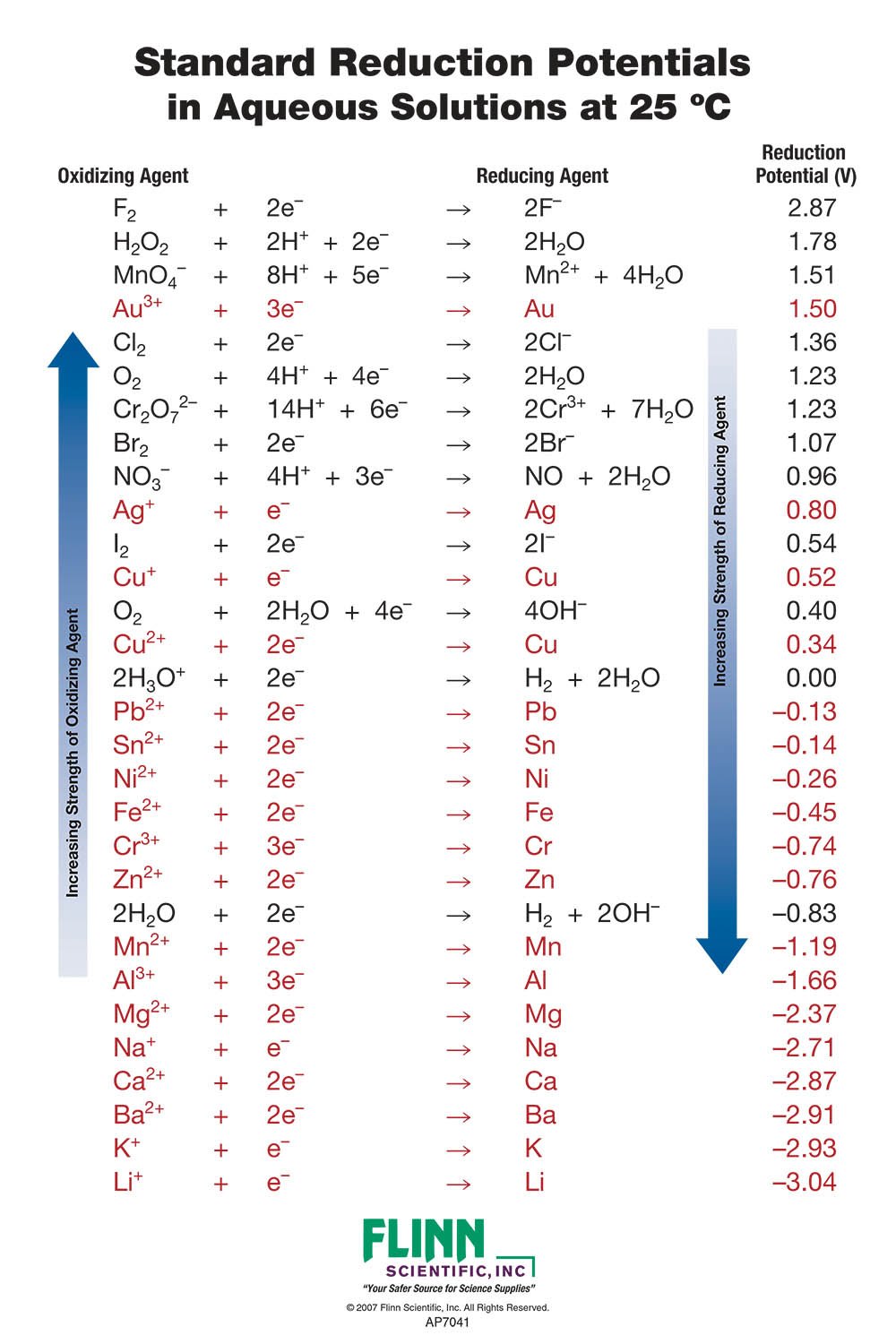
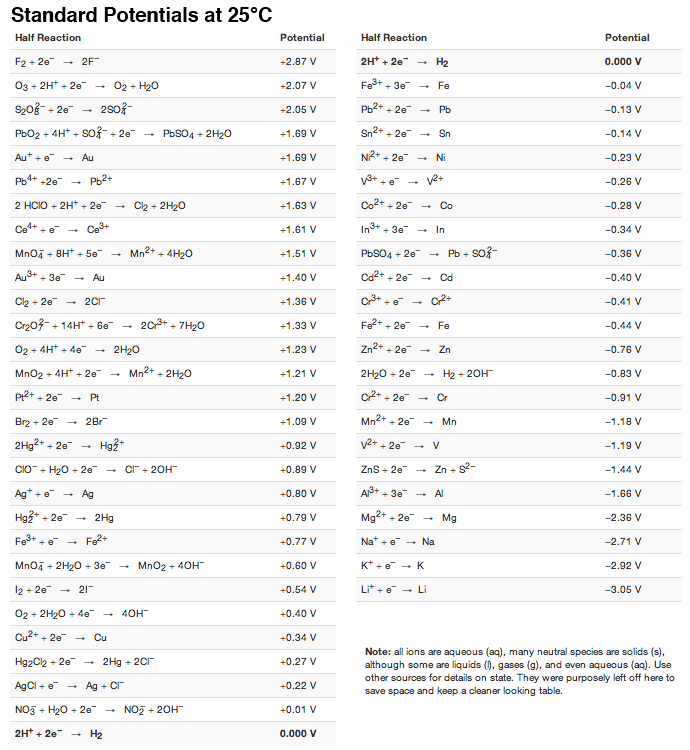
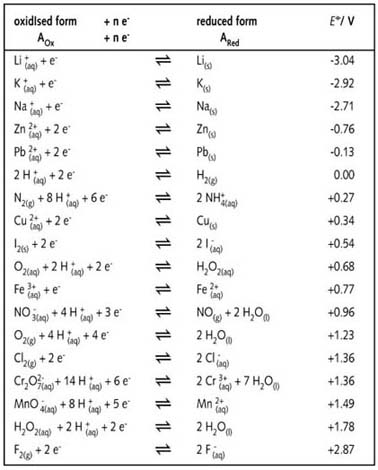
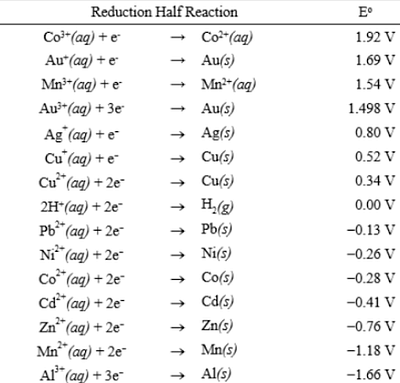
Using the standard electrode potentials given in Table 3.1, predict the reaction between the following is feasible:(i) Fe^{3+}(aq) and I^{-}(aq)(ii) Ag^{+} (aq) and Cu(s)(iii) Fe^{3+} (aq) and Br^{-} (aq)(iv) Ag(s) and Fe^{3+} (

Table of Standard Reduction Potentials and Activity Series.pdf - Table of Standard Reduction Potentials Cathode Reduction Half Reaction | Course Hero

Using the standard electrode potentials given in the table, predict the reaction between the following is possible.Fe^{3+}(aq) and I^{-}(aq)

Table 4 from Estimation of standard reduction potentials of halogen atoms and alkyl halides. | Semantic Scholar
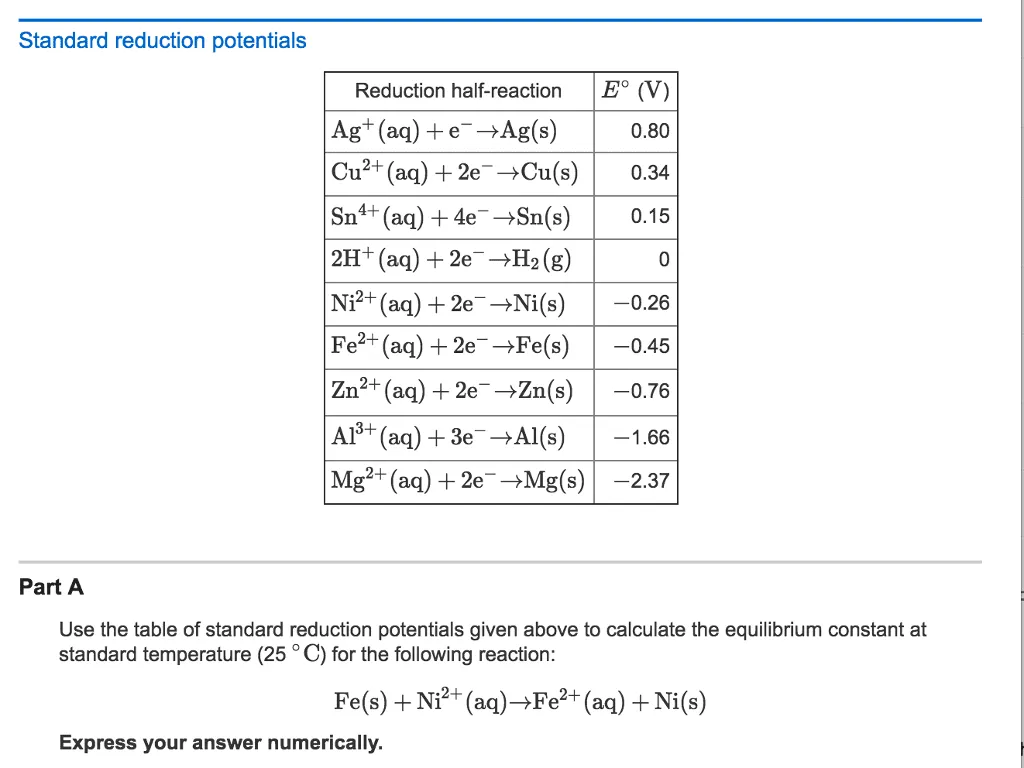
OneClass: Standard reduction potentials Use the table of standard reduction potentials given above to...
![PDF] Standard Electrode Potentials and Temperature Coefficients in Water at 298.15 K | Semantic Scholar PDF] Standard Electrode Potentials and Temperature Coefficients in Water at 298.15 K | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/56964684a624c5af38c7e62256db3faa4c542d88/19-Table2-1.png)
PDF] Standard Electrode Potentials and Temperature Coefficients in Water at 298.15 K | Semantic Scholar

SOLVED: Table A5.5 Standard Reduction Potentials at 25°C (298 K) for Many Common Half-Reactions Half-Reaction Half-Reaction 4OH- + F2 â†' 2F- + 2H2O 2.87 2H2O + 4e- â†' Cu2+ + 2OH- 0.01